
The same notation can also be found in the IUPAC Green Book Quantities, Units and Symbols in Physical Chemistry (2007). The mass, charge and atomic number of a nuclide are indicated by means of three indexes (subscripts and superscripts) placed around the symbol. Note, however, that this document unfortunately introduced the terrible typographic disaster of the staggered notation for ions (see this question). This notation is also used in Nomenclature of Inorganic Chemistry – IUPAC Recommendations 2005 (Red Book). The same meanings are described in the German standard DIN 1338 (2011). Long story short, the second notation $(\ce$$ Books are more trustworthy as long as they are written by scientists. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). Block Elements are organised into blocks by the orbital type in which the outer electrons are found. IUPAC votes to change standard atomic weights of 19 elements.Periodic tables of elements (PTEs) are often abused by designers. The atomic number of each element increases by one, reading from left to right. 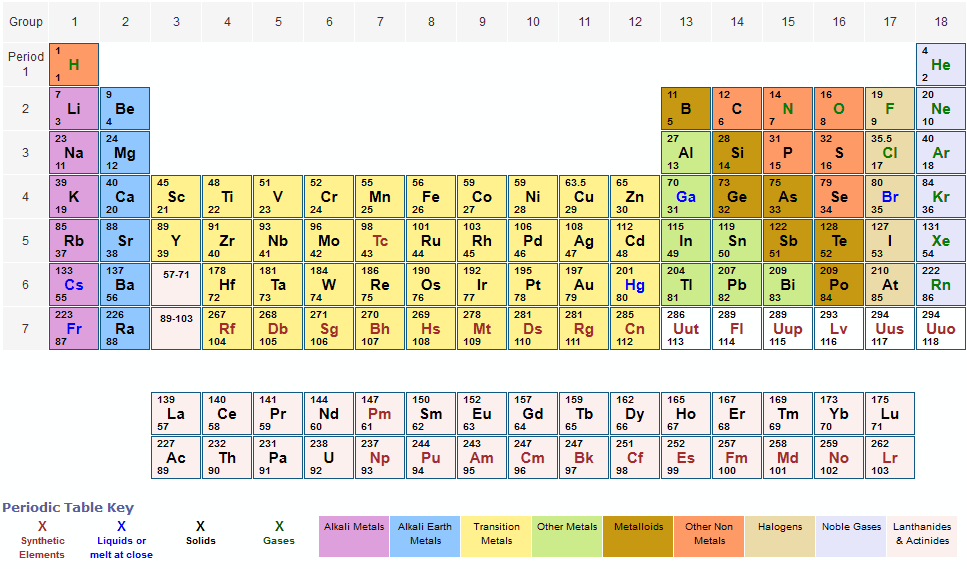
Retrieved from /39912-atomic-weight-changed-for-19-elements.html The changes will be published in a new “Table of Standard Atomic Weights 2013” in Pure and Applied Chemistry in 2014 as follows: Besides, this change could potentially affect you as well since the gold bar you own might be worth less now due to the slight increase in gold’s atomic mass! Though these changes may seem subtle and you may wonder why the chemists have gone through so much hassle, but in fact, new weights could tremendously impact scientific research and many other fields that rely on chemistry. The number in parentheses represents the uncertainty in the last digit of the atomic weight. The weight of gold, for example, has been updated from 196.966 569 (4) amu (atomic mass unit) to 196.966 569 (5) amu. For the remaining elements, the new weights were determined through more precise measurements and only minor changes have been made. The modification in weight change for the non-metal selenium is particularly remarkable as it has not been revised since 1934. The standard atomic weights of four elements-molybdenum, cadmium, selenium, and thorium have been changed based on recent determinations of terrestrial isotopic abundances. 
The diagram further explains how the atomic weight is calculated. Chemists calculate the average atomic mass of an element that you see on the periodic table from the masses of its isotopes, giving more common isotopes more weight than less common isotopes. In the case of carbon, the most abundant stable isotopes can be found as carbon-12 and carbon-13. These different versions of element’s atoms that have the same number of protons but different numbers of neutrons are called isotopes. However, each carbon atom also has neutrons in its nucleus, and different types of carbon atoms exist that have different numbers of neutrons. Thus, a carbon atom has six electrons orbiting around its nucleus. For the atoms to be neutral (no net charge), the number of electrons must equal the number of protons. How do we calculate the atomic weight? To use carbon as an example, a carbon atom has six protons in its nucleus. Scientists have been publishing tables with atomic weights that were determined as far back as 1899. IUPAC has approved new weights for these elements due to better measurements and calculations of certain isotopes. 
Wow Cadmium, you have gotten chubbier these days!Īccording to the International Union of Pure and Applied Chemistry (IUPAC), the federation responsible for overseeing international standards for atomic weights since 1919, nineteen elements on the periodic table are getting their atomic weight adjusted.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
